Live Case Presentations During Investigational Device Exemption (IDE) Clinical Trials - Guidance for Institutional Review Boards
FDA Categorization of Investigational Device Exemption (IDE) Devices to Assist the Centers for Medicare and Medicaid Services (C

FDA Regulation of Neurological and Physical Medicine Devices: Access to Safe and Effective Neurotechnologies for All Americans - ScienceDirect
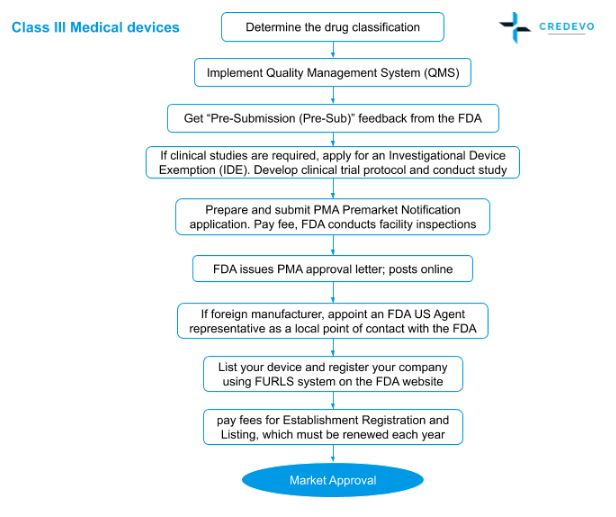
Page 1 of 5 Title: Requirements for Investigational Device Exemption (IDE) for Human Subject Research Department: Human Research
Guidance and Procedures: Use of Devices in Clinical Research and Brief Overview Investigational Device Exemption Applications (

Updated FDA Guidance Helps Device Study Sponsors Better Anticipate Coverage for Investigational Devices | Advisories | Arnold & Porter
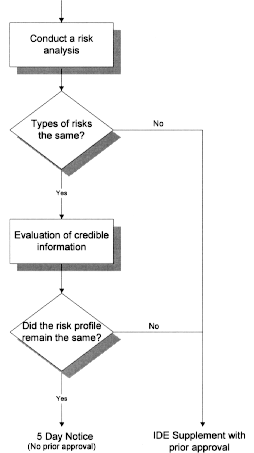
Changes or Modifications During the Conduct of a Clinical Investigation; Final Guidance for Industry and CDRH Staff | FDA
FDA Decisions for Investigational Device Exemption Clinical Investigations - Guidance for Sponsors, Clinical Investigators, Inst
Wednesday June 2, 2016 FDA Categorization of Investigational Device Exemption (IDE) Devices to Assist the Centers for Medicare
FDA Categorization of Investigational Device Exemption (IDE) Devices to Assist the Centers for Medicare and Medicaid Services (C